A new study published in the Proceedings of the National Academy of Sciences (PNAS) reveals that organized civil society and social mobilizations are key, yet often unrecognized, agents of global biodiversity conservation.
Author Archive
A new study refines the dating of human past on the Cantabrian coast 18,000 years ago
A new study refines radiocarbon dating of marine remains and significantly improves the precision with which the human past of the Magdalenian period in the Cantabrian region of Spain can be reconstructed, a key phase of European prehistory dating to around 18,000 years ago.
Microscopic plankton reveal tropicalization of the Mediterranean Sea
The Mediterranean Sea is rapidly changing under ongoing climate change. In the eastern basin, tropicalization is already well documented and driven by a combination of strong warming and the influx of tropical species through the Suez Canal.
Fossil fuel industry’s “false climate solutions” reinforce its power and aggravate environmental injustice
Many so-called low-carbon projects promoted by major oil and gas companies — including hydrogen, biofuels, carbon capture and storage, and carbon offsetting — operate as false solutions that not only fail to effectively reduce emissions, but also prolong the lifespan of fossil fuel infrastructures, entrench environmental injustices, and reinforce the political and economic power of the very industry responsible for the climate crisis.
Bolivia at a crossroads: scientists warn of a possible environmental setback
Bolivia, one of the most biodiverse countries on the planet, stands at a critical juncture.
Happy Holidays!
From ICTA-UAB, we wish you Happy Holidays and a Happy New Year 2026.
Thank you for sharing with us, throughout this year, your ideas, experiences, collaboration and commitment, all of which continue to inspire our work.
We look forward to continuing this journey together, growing and strengthening Brave Science For A Changing World, and facing together with hope, enthusiasm and care the major societal challenges arising from environmental issues.
Whale hunting in South America began 5,000 years ago, a millennium earlier than previously thought
The hunting of large whales goes back much further in time than previously thought. New research from ICTA-UAB and the Department of Prehistory of the UAB reveals that Indigenous communities in southern Brazil were hunting large cetaceans 5,000 years ago, around a thousand years before the earliest documented evidence from Arctic and North Pacific societies.
Preparación temporal: ¿Cómo anticipamos el momento justo?

En un estudio reciente, un grupo de investigadores ha creado la herramienta Temporal Preparation Task (TEP-Task). En esta tarea se miden diferentes efectos de preparación temporal en bloques separados, mientras que todo lo demás permanece constante: los mismos estímulos, el mismo procedimiento y el mismo entorno.
Seguir leyendo
Temporal Preparation: How Do We Anticipate the Right Time?

In a recent study, a group of researchers create the tool Temporal Preparation Task (TEP-Task). In this task, different temporal preparation effects are measured in separate blocks, while everything else remains constant: the same stimuli, the same procedure and the same setting.
Seguir leyendo
Nature Metabolism: A yeast enzyme helps human cells overcome mitochondrial defects[
Nucleotide synthesis—the production of the basic components of DNA and RNA—is essential for cell growth and division. In most animal cells, this process depends closely on properly functioning mitochondria, the organelles responsible for respiration and energy production. When mitochondrial respiration fails—a common feature of mitochondrial diseases and several forms of cancer—cells lose the ability to proliferate normally. A new study published in Nature Metabolism now shows that this dependence is not irreversible.
An international team led by José Antonio Enríquez of the Centro Nacional de Investigaciones Cardiovasculares Carlos III (CNIC) and the Spanish network for research into frailty and healthy aging (CIBERFES) has experimentally uncoupled nucleotide synthesis from mitochondrial activity using ScURA, a yeast-derived genetic tool now available to the research community that will enable new explorations of cellular metabolism.
The study, which also involved scientists at the University of Cologne (Germany), the University of Valladolid (UVa), and the CSIC–UVa Institute of Biology and Molecular Genetics, sheds new light on the role of mitochondria in rare diseases and cancer.
In complex organisms such as humans, respiration is essential for generating the energy required for life, with the mitochondria in our cells using oxygen to sustain vital cellular processes. By contrast, some organisms—such as the yeast Saccharomyces cerevisiae—can survive without oxygen and have evolved alternative metabolic pathways to produce the molecular building blocks required for RNA and DNA synthesis.
Building on this observation, the team identified a yeast enzyme that can sustain nucleotide synthesis independently of mitochondrial respiration. Instead of oxygen, this enzyme uses fumarate, a metabolite derived from nutrients. The team extracted the gene encoding this enzyme, called ScURA, from yeast and inserted it into human cells.
Unlike cells from healthy individuals, the patient-derived cells used in the study cannot grow in standard laboratory conditions because they require extra supplementation with nutrients and DNA precursors. When CNIC researchers introduced ScURA into these diseased cells, they found that the cells were able to grow under normal conditions, just like cells from healthy individuals. “Thanks to the yeast gene, the cells ‘learned’ to build DNA in a new way,” explain the authors.
The results were striking: human cells expressing ScURA continued to produce DNA and RNA even when the mitochondrial respiratory chain was blocked. Unlike the equivalent human enzyme, which is physically linked to the mitochondria, the yeast version works in the cytosol and uses an alternative metabolic pathway.
The team also discovered that ScURA helped cells use their nutrients more efficiently without disrupting other essential cellular functions—an important first step toward the more ambitious goal of improving the lives of people with mitochondrial disorders.
“Mitochondria not only produce energy; they also shape fundamental processes such as DNA synthesis,” says lead author José Antonio Enríquez, head of the CNIC GENOXPHOS group. “Our work shows that if we provide a cell with an alternative route to make nucleotides, we can sustain cell proliferation even when mitochondrial respiration fails.”
One of the study’s most important findings is that ScURA-modified cells can grow without uridine supplementation, a common strategy used in laboratories to compensate for mitochondrial defects.
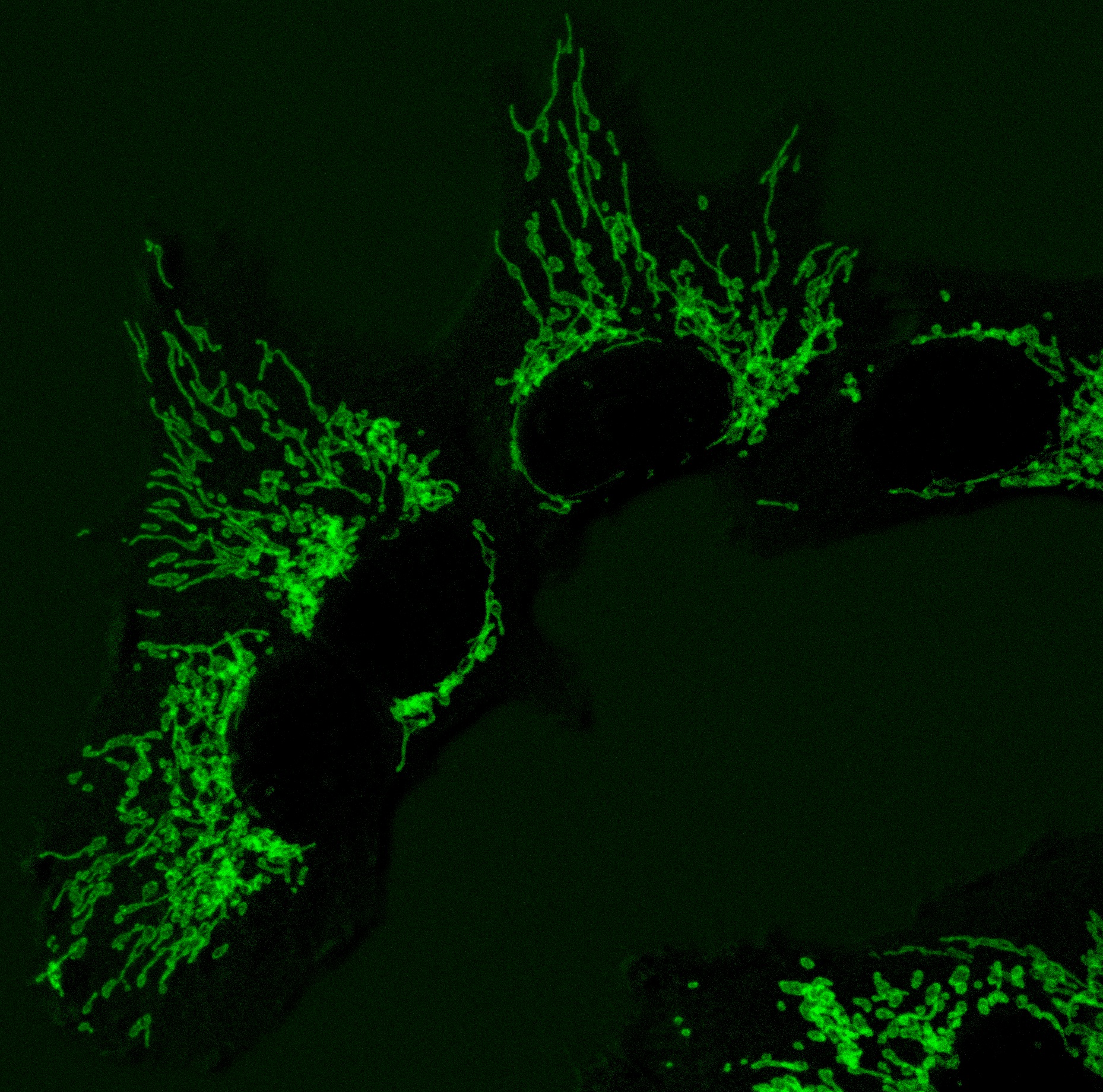
Moreover, the new approach restores cell proliferation across different experimental models of mitochondrial diseases, including those caused by severe mutations in essential respiratory chain complexes. For first author Andrea Curtabbi (CNIC), “this tool allows us, for the first time, to clearly separate the direct effects of mitochondrial dysfunction on nucleotide synthesis from other secondary metabolic changes.”
Mitochondrial diseases are severe and often untreatable, and in the laboratory cells with impaired mitochondrial respiration require external supplements to proliferate. However, when the researchers inserted ScURA into these cells, they proliferated under standard conditions in the same way as healthy cells.
The study also shows that this imported enzyme boosts the efficiency of nutrient use without altering other essential cellular functions, making ScURA a highly valuable experimental tool. The authors further highlight its potential for clarifying mitochondrial contributions to rare diseases and cancer. “Identifying which metabolic processes become limiting when mitochondrial respiration fails is crucial for designing precise therapeutic strategies,” concludes Enríquez.
In future work, the team plans to expand their findings to other disease models and optimize this approach for preclinical research.
The project was funded by the Spanish Ministry of Science and Innovation (projects PID2021-127988OB-I00 and TED2021-131611B-100), the Human Frontier Science Program (RGP0016/2018), the Leducq Foundation (17CVD04), and the Instituto de Salud Carlos III – CIBERFES (CB16/10/00282).
ICTA-UAB participates with ONAC in 204 climate proposals
Researchers from the ICTA-UAB have actively participated in the drafting process of the Working Document on Proposals from the Scientific Community in Response to the Climate Emergency, promoted by the Spanish National Office for Scientific Advice (ONAC), a Government of Spain body responsible for integrating scientific knowledge into public policymaking.
Genomic Analysis in Neuroscience Course, 2nd edition – October 2026
Genomic Analysis in Neuroscience Course, 2nd edition
October 2026 — Full programme and registration details coming soon.
La entrada Genomic Analysis in Neuroscience Course, 2nd edition – October 2026 se publicó primero en Instituto de Neurociencias de Alicante.
